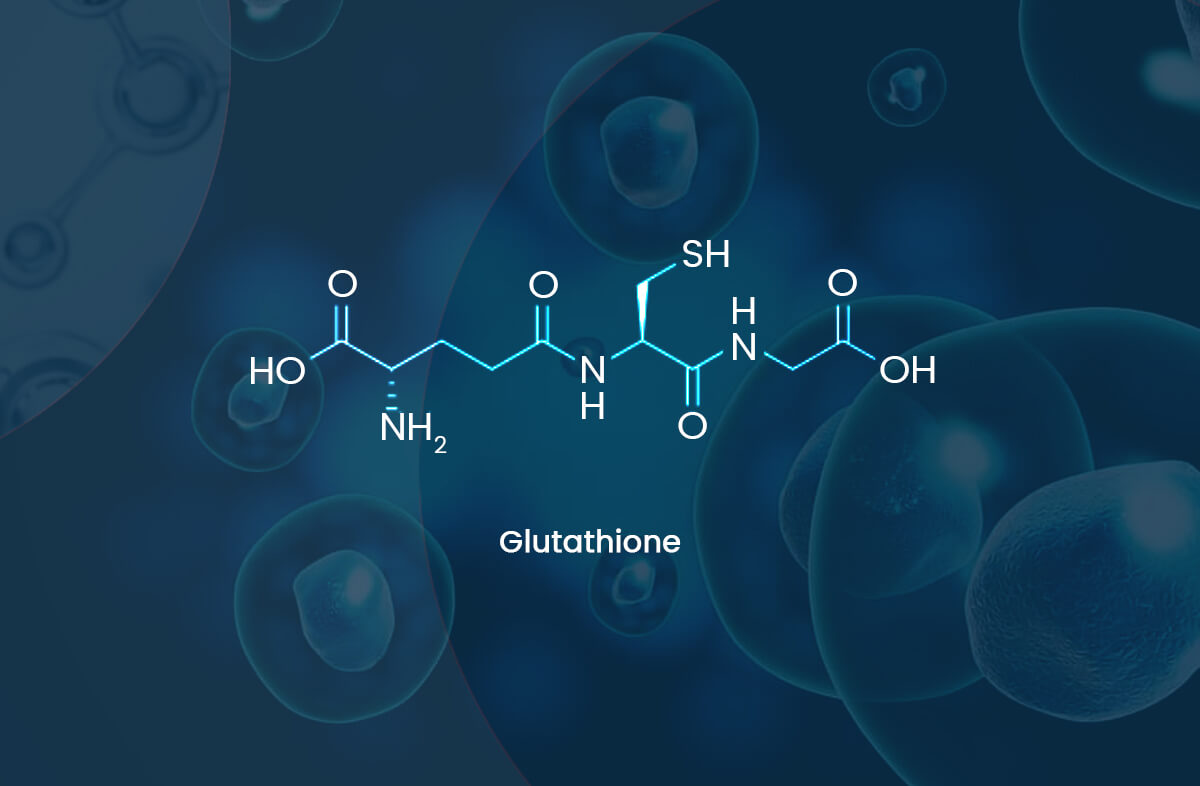
Glutathione (GSH) is synthesized in the cytoplasm in virtually all cells from its constituent amino acids by two sequential ATP-requiring enzyme catalyzed reactions (see figure below) [1]. The first reaction is the formation of the dipeptide γ-glutamylcysteine (GGC) from the amino acids L-glutamic acid and L-cysteine. The second reaction adds a glycine to the Gamma-glutamylcysteine to produce the tripeptide glutathione.
The first reaction is rate limiting and is catalyzed by the enzyme glutamate-cysteine ligase (GCL, EC 6.3.2.2; formerly γ-glutamylcysteine synthetase). GCL is composed of a heavy catalytic subunit (GCLC, MW ~ 73,000) and a light modifier (GCLM, MW ~ 30,000) subunit. GCL is the key control point for the homeostasis of cellular GSH and is regulated at multiple levels. Its regulation at a genetic level (both transcriptional and translational) and at a biochemical level (post-translational) are incredibly complex.
The light modifier subunit GCLM is enzymatically inactive but plays an important regulatory function by lowering the Km of GCLC for glutamate and raising the Ki for glutathione (GSH) [7, 8]. The resulting holoenzyme dimer comprising the two subunits is catalytically more efficient and less subject to inhibition by glutathione (GSH) than GCLC alone. In rats, GCLC has a Km for glutamate that is about 10-fold higher than that of the GCL holoenzyme, which is higher than the cellular glutamate concentration in most tissues
GCL is specific for the glutamyl moiety and is regulated physiologically by: (a) non-allosteric feedback competitive inhibition by glutathione (GSH) (Ki = 2.3 mM) which involves binding of GSH to the glutamate and another site on the enzyme [8, 9] and (b) availability of its precursor, cysteine [1]. The apparent Km values of GCL for glutamate and cysteine are 1.8 and 0.1–0.3 mM, respectively [9]. The intracellular glutamate concentration is nearly 10-fold higher than the Km value, but intracellular cysteine concentrations approximate the apparent Km value [10] indicating that cysteine may be a limiting substrate for GCL. This has led to the false assumption that the cause of glutathione (GSH) depletion associated with so many chronic diseases must be from an under supply of cysteine in the diet. This is an unlikely scenario as the developed world dietary intake of sulfur amino acids is generally adequate. For example, the typical American diet supplies much more than the recommended required quantity of cysteine [11]. Interestingly, this has not discouraged multiple research groups testing cysteine prodrugs, such as N-acetylcysteine (NAC), as supplements to increase glutathione (GSH) levels. Unsurprisingly, most clinical trials with NAC have demonstrated a lack of efficacy [12-16]. However, NAC has been shown to restore glutathione levels in cases such as acetaminophen overdose where an acute depletion of glutathione is observed [17]. In these cases, glutathione (GSH) levels are well below the normal concentration and therefore feedback inhibition of GCL is absent. To this date, the only medically recognized use of NAC in increasing cellular glutathione is in acetaminophen poisoning.
Our proposition is that the suboptimal glutathione levels associated with so many chronic diseases are not due to a cysteine undersupply but an impairment in one of the multiple control mechanisms of the GCL enzyme. Again, this impairment can be effectively bypassed by supplementation with gamma-glutamylcysteine (GGC) which can be transported into cells where it will be rapidly converted to glutathione by the enzyme Glutathione Synthase (GS).
The second step in glutathione (GSH) synthesis is catalyzed by the enzyme Glutathione Synthase (GS, EC 6.3.2.3, formerly known as GSH synthetase). GS is constitutively expressed in all cells and has not been studied as extensively as GCL [5]. It is not subject to feedback inhibition by glutathione (GSH) and its substrates gamma-glutamylcysteine (GGC) and glycine are rapidly ligated to form glutathione (GSH). The intracellular concentration of gamma-glutamylcysteine (GGC) is extremely low (~ 5 uM) when GS is present, thus GCL is considered the rate-limiting enzyme [6]. In support of this observation, over expression of GS in a yeast model failed to increase glutathione (GSH) levels whereas over expression of GCL did [18].
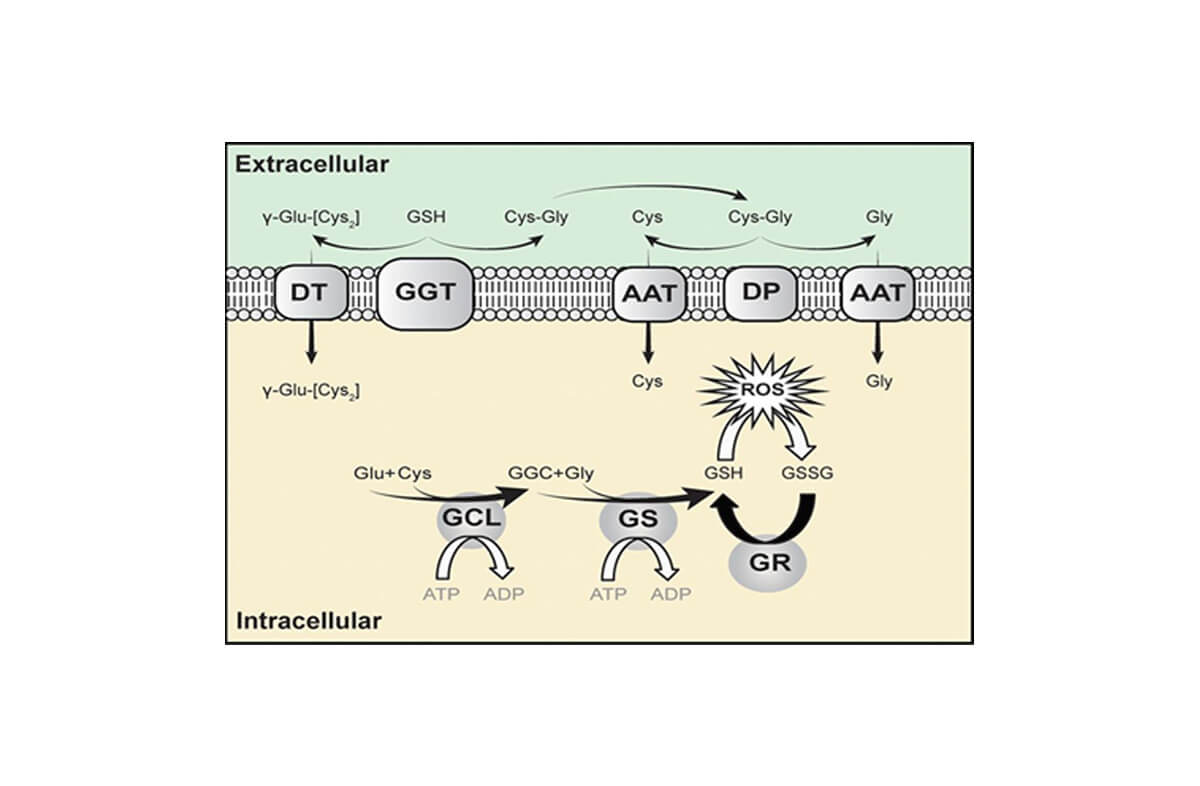
The intracellular concentration of glutathione (GSH) is in the millimolar range and the extracellular concentration is in the micromolar range. This steep glutathione (GSH) concentration gradient from inside the cell to outside the cell drives the export of glutathione (GSH) from the cell. This concentration gradient of glutathione (GSH) makes transport of glutathione (GSH) from outside the cell to inside the cell thermodynamically unfavorable. It is not surprising then that most attempts to increase intracellular glutathione (GSH) by glutathione supplementation have been unsuccessful [19, 20]. Once outside the cell, glutathione (GSH) degradation occurs exclusively in the extracellular space, and only on the surface of cells that express the ectoenzyme γ-glutamyl transferase. This enzyme catalyzes the transfer of the γ-glutamyl moiety of glutathione (GSH) to another amino acid to produce a γ-Glu-AA and cysteinyl-glycine. These γ-Glu-AAs can be transported back into cells, where they become substrates for the enzyme γ-glutamyl cyclotransferase. This enzyme generates 5-oxoproline and releases the amino acid or peptide that is bound to glutamate. 5-Oxoproline is converted to glutamate by the ATP-requiring enzyme 5-oxoprolinase. The other part of the hydrolyzed glutathione (GSH) molecule cysteinyl-glycine is transported back inside the cell and broken down further by nonspecific dipeptidases into cysteine and glycine ready providing substrates for glutathione (GSH) resynthesis. This process allows for the release of cysteine from glutathione (GSH) which can then be used in protein synthesis. In this case, glutathione (GSH) is functioning as a storage mechanism for cysteine which is otherwise unstable in its free form.
Glutathione (GSH) is a cofactor, coenzyme, and/or substrate for a number of enzymes, and can participate in a number of redox and conjugation reactions. Within the cell, it exists mainly (98%) in the thiol-reduced form glutathione (GSH), but some is also present as glutathione disulfide (GSSG). This ratio of GSH:GSSG is maintained by the action of the enzyme GSSG reductase (also known as glutathione reductase) which requires NADPH+ as the reducing cofactor. Glutathione (GSH) can react with many electrophilic compounds to generate glutathione S-conjugates. Free radicals are neutralized and other oxidants are reduced by the direct action of glutathione (GSH) and indirectly via enzyme glutathione peroxidase, which used glutathione (GSH) as a cofactor.
References
- Meister, A. and M.E. Anderson, Glutathione. Annu Rev Biochem, 1983. 52: p. 711-60.
- Ferguson, G. and W. Bridge, Glutamate cysteine ligase and the age-related decline in cellular glutathione: The therapeutic potential of γ-glutamylcysteine. Archives of Biochemistry and Biophysics, 2016. 593: p. 12-23.
- Franklin, C.C., et al., Structure, function, and post-translational regulation of the catalytic and modifier subunits of glutamate cysteine ligase. Molecular Aspects of Medicine, 2009. 30(1-2): p. 86-98.
- Ballatori, N., et al., Glutathione dysregulation and the etiology and progression of human diseases. Biological Chemistry, 2009. 390(3): p. 191-214.
- Lu, S.C., Glutathione synthesis. Biochimica et Biophysica Acta (BBA) – General Subjects, 2013. 1830(5): p. 3143-3153.
- Dalton, T.P., et al., Genetically altered mice to evaluate glutathione homeostasis in health and disease. Free Radical Biology and Medicine, 2004. 37(10): p. 1511-1526.
- Huang, C.S., M.E. Anderson, and A. Meister, Amino acid sequence and function of the light subunit of rat kidney gamma-glutamylcysteine synthetase. J Biol Chem, 1993. 268(27): p. 20578-83.
- Huang, C.S., et al., Catalytic and regulatory properties of the heavy subunit of rat kidney gamma-glutamylcysteine synthetase. J Biol Chem, 1993. 268(26): p. 19675-80.
- Richman, P.G. and A. Meister, Regulation of gamma-glutamyl-cysteine synthetase by nonallosteric feedback inhibition by glutathione. J Biol Chem, 1975. 250(4): p. 1422-6.
- Bannai, S. and N. Tateishi, Role of membrane transport in metabolism and function of glutathione in mammals. J Membr Biol, 1986. 89(1): p. 1-8.
- Lang, C.A., The impact of glutathione on health and longevity. Journal of Anti Aging Medicine, 2001. 4(2): p. 137-144.
- Aitio, M.-L., N-acetylcysteine – passe-partout or much ado about nothing? British Journal of Clinical Pharmacology, 2006. 61(1): p. 5-15.
- Ashworth, A. and S.T. Webb, Does the prophylactic administration of N-acetylcysteine prevent acute kidney injury following cardiac surgery? Interact CardioVasc Thorac Surg, 2010. 11(3): p. 303-308.
- Sochman, J., N-Acetylcysteine Somewhere Between Scylla and Charybdis. J Am Coll Cardiol, 2010. 56(13): p. 1067-a-.
- Wang, G., et al., N-acetylcysteine in Cardiac Surgery: Do the Benefits Outweigh the Risks? A Meta-Analytic Reappraisal. Journal of cardiothoracic and vascular anesthesia, 2011. 25(2): p. 268-275.
- Coles, L.D., et al., Repeated-Dose Oral N-Acetylcysteine in Parkinson’s Disease: Pharmacokinetics and Effect on Brain Glutathione and Oxidative Stress. The Journal of Clinical Pharmacology, 2018. 58(2): p. 158-167.
- Rushworth, G.F. and I.L. Megson, Existing and potential therapeutic uses for N-acetylcysteine: The need for conversion to intracellular glutathione for antioxidant benefits. Pharmacology & Therapeutics, 2014. 141(2): p. 150-159.
- Grant, C.M., F.H. MacIver, and I.W. Dawes, Glutathione synthetase is dispensable for growth under both normal and oxidative stress conditions in the yeast Saccharomyces cerevisiae due to an accumulation of the dipeptide gamma-glutamylcysteine. Mol Biol Cell, 1997. 8(9): p. 1699-707.
- Witschi, A., et al., The systemic availability of oral glutathione. European Journal of Clinical Pharmacology, 1992. 43(6): p. 667-669.
- Allen, J. and R.D. Bradley, Effects of oral glutathione supplementation on systemic oxidative stress biomarkers in human volunteers. J Altern Complement Med, 2011. 17(9): p. 827-33.